21 CFR Part 211 Guide
This app provides the full text of FDA’s 21 CFR Part 211 regulation for current Good Manufacturing Practice. The app is provided as a handy, easily available mobile reference for practitioners of the regulation, who can access the content of the regulation at any time, no matter where they are.
Features:
- Navigate text using the Table of Contents
- Search content using keywords
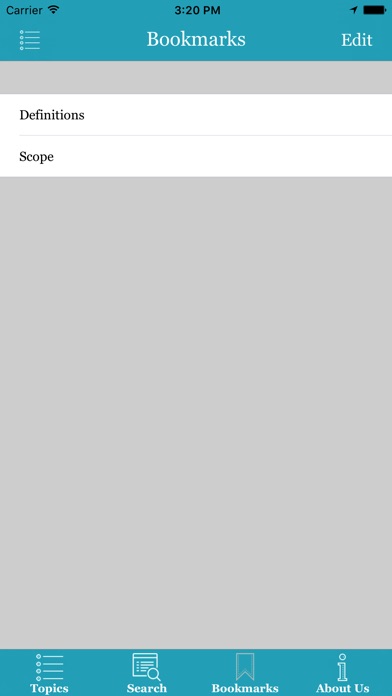
- Bookmark pages of interest
- Email a page to yourself or a colleague
This app is provided by CIMINFO Software, Inc., a leading provider of 21 Part 11 compliant software and solutions to the FDA-regulated industry. CIMINFO provides software solutions to manage spreadsheets, documents, SOPs, databases, records, drawings, manufacturing equipment/assets, and training records. For more information, please visit www.part11solutions.com or email us at info@ciminfo.net.